What changed the world around
1900--created the new "modern" world view? it took
more than changing technology
Modernism meant seeing the world in a new way that science reinforced:

people become more willing to throw out old ways and try to come up with completely new ways of doing things
We will come back to art, but next we will consider inventors and scientists
- a world
view is the set of assumptions we have about how the
world works
- technological change changed the world so much by the
1890s that people began to expect new ways of seeing the
world to go with that new way of life
- radical changes in science changed people's sense of the world
- modern art is a cause as well as an effect
relationship between new ideas in science and
technology:
- the new physics led to people being more willing to throw out tradition in other areas
- new technology is changing the world inspiring scientists to try new ideas
- new science led to some new technologies
- today: science keeps changing its mind (increased
information)
- today: science is an established authority that people reject (increased individualism)
- around 1900: more enthusiasm for science and other kinds of progress
- but science was also changing in disorienting ways
- it became clear that a revolutionary change in physics was underway
- in traditional physics, things were either particles
or waves, Newton had shown that light was waves
- physicist Max Planck published a scientific paper showing that energy is not continuously distributed like a wave but divide up into quanta, which are more like particles--this was a big shock in physics and led to the work of Einstein and to quantum mechanics
- physics starts showing things about the world that are totally weird--this shook up educated people. If science has showed the world is fundamentally different from our common sense, then it is easier to throw out common sense in other areas
- common sense: gut feeling that something is right,
but it comes from the people around us and social norms
- new theories went in surprising directions but also
led to new technologies
- x-rays caught the imagination of the public--can you imagine for the first time being able to see though flesh?
- science was shaking up the stable world people were
used to
- begin to get examples of new science leading to new
technology within 20 years--the frontier of science became
more connected with the experience of everyday people
Consider more carefully the
new view of the atom: video
- the word atom was supposed to mean the smallest piece, of which everything is made
- since the late 1700s, chemists had a fairly clear
theory that matter was made of combinations of atoms of
a limited number of elements,
- the discovery of elements suggested that atoms of the different elements were the basic building blocks of everything (the first scientific discovery of an element was in 1649)
- but what was an atom? A tiny bowling ball? The revolution was a new theory--Quantum mechanics--showed atoms were extremely complicated and didn't behave in the ways larger objects did.
- Newton thought light was made up of particles but
research in the 1820s showed that light behaves like a
wave
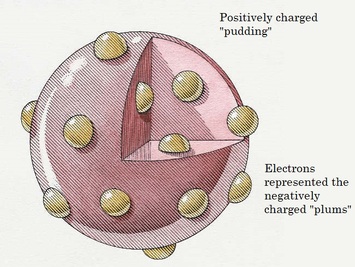
- in 1897 J. J. Thompson discovered the electron (by seeing that electric current could pass through the vacuum in a vacuum tube)--the atom is not a bowling ball but has a positively charged part and a negatively charged part that can be separated. Thompson thought that atoms looked like raisin bread (or in England they said like a plum pudding), with electrons embedded in them
- in
1901 Planck proved theoretically that energy is
emitted in units (quanta), not as a continuous
wave. This could not be explained within the
classical theory of physics, so Planck concluded that
the laws of classical physics do not apply to atoms
- in 1905 Einstein published three important papers
- one showed that the photoelectric effect (where light hitting certain materials will create an electrical current) behaves in such a way that light must be particles with particular energy, not continuous waves
- the second paper was the special
theory of relativity--time moves slower as you
get closer to the speed of light
- the third explained Brownian
motion, providing evidence that atoms really
existed
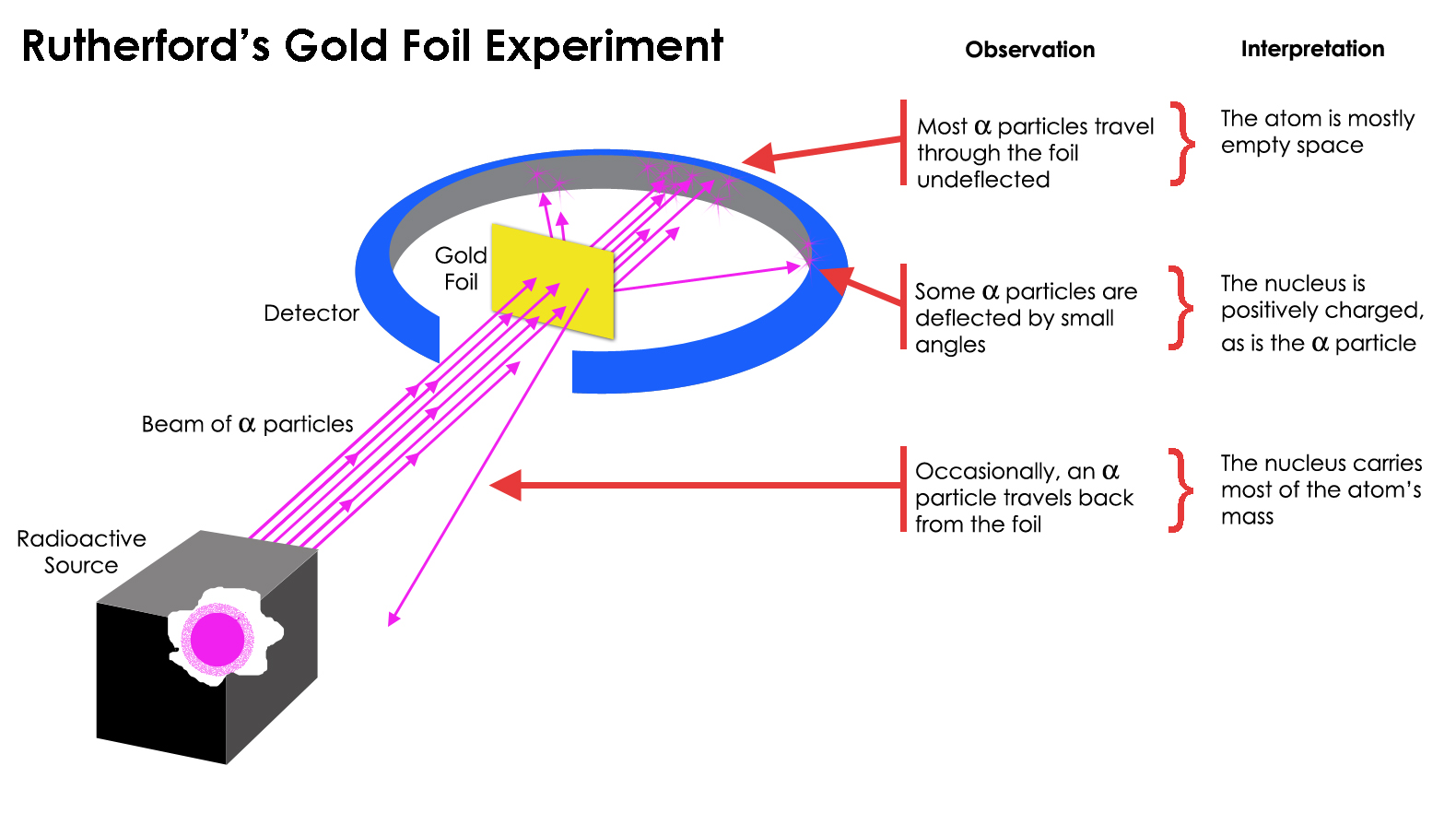
- In 1908 Ernst Rutherford did an experiment where he
beamed alpha particles at a piece of very thin gold
foil. Alpha particles were known to be very small atoms
(actually the nucleus of the helium atom) and gold was
made up of big atoms. Most of the alpha particles
passed through the foil, but some
bounced back. The shocking
conclusion was that most of the atom was empty
space but part of it was very dense--the plum pudding
(raisin bread) model was wrong.
- This led Rutherford to propose in 1911 the idea that the atom looked something like the solar system, with a small dense positively-charged nucleus with electrons swirling around it.
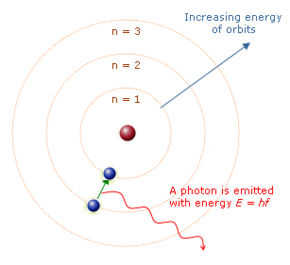
- in 1913 Niels Bohr proposed that the electrons can only travel in fixed orbits around the nucleus, so when electrons move from a higher orbit to a lower one they emit radiation with a fixed amount of energy.
- in 1924 de Broglie theorized that not only light but also electrons and other subatomic particles have the properties of particles and the properties of waves at the same time
- in 1925 Werner Heisenberg put together the pieces
of quantum mechanics. This required the idea that
an electron moved
from one orbit to another orbit without passing
through the space in between as well as the uncertainty
principle (below)
Modernism meant seeing the world in a new way that science reinforced:

- matter was no longer solid--we could see through it
- science isn't going to be straightforward and commonsense and reassuring any more:
"Nature and Nature's Laws
lay hid in Night/
God said, Let Newton be! and all was Light." -- Alexander Pope.
"It did not last; the Devil howling 'Ho!
Let Einstein Be!' restored the status quo." - John Collings Squire
God said, Let Newton be! and all was Light." -- Alexander Pope.
"It did not last; the Devil howling 'Ho!
Let Einstein Be!' restored the status quo." - John Collings Squire
- Heisenberg Uncertainty Principle--we cannot observe an atom without affecting it--until then its position and movement cannot be known for sure. Schrodinger's cat is an illustration of this
- because the components of matter and energy have the characteristics of both particles and waves at the same time, they simply don't have a fixed state or position
- Special Relativity: time moves more slowly when you move close to the speed of light
- pointillism
and cubism
in art

- our view of reality fragmented--there was no longer a nice stable view of the world that everyone agreed on
- atonality in music (and John Cage's 4'33")
- classical music and any music based on playing
chords (as on a guitar or piano) is based on a
traditional system of harmony. Composers in the modern
era began to experiment with throwing out the whole
traditional system and trying new combinations of notes
that didn't follow the patterns people are used to
hearing
- Joyce's Ulysses was particularly famous as a radical
book throwing out the traditions of how a novel should
work and replacing them with stream of consciousness
people become more willing to throw out old ways and try to come up with completely new ways of doing things
We will come back to art, but next we will consider inventors and scientists